The disclosure-dedication doctrine applies at limitation level, not at embodiment level
| May 27, 2020
Eagle Pharmaceutical Inc. v. Slayback Pharm LLC
May 8, 2020
O’Malley (Opinion author), Reyna, and Chen
Summary
The Federal Circuit affirmed the district court’s holding that the disclosure-dedication doctrine applies here to bar application of the doctrine of equivalents and, accordingly, the Slayback’s generic products do not infringe the Eagle’s four patents.
Details
Slayback filed new drug application (“NDA”) for a generic version of Eagle’s branded bendamustine product, BELRAPZO®. Bendamustine is used to treat chronic lymphocytic leukemia and indolent B-cell non-Hodgkin lymphoma. Eagle sued Slayback for infringing four patents in the District Court for the District of Delaware. Eagle’s four asserted patents share essentially the same written description. One of the four patents is U.S. Patent No. 9,572,796 (“the ’796 patent”), which has a representative claim 1 as shown below:
1. A non-aqueous liquid composition comprising: bendamustine, or a pharmaceutically acceptable salt thereof;
a pharmaceutically acceptable fluid comprising a mixture of polyethylene glycol and propylene glycol, wherein the ratio of polyethylene glycol to propylene glycol in the pharmaceutically acceptable fluid is from about 95:5 to about 50:50; and
a stabilizing amount of an antioxidant;
. . . .
In the district court, Slayback conceded that its generic product literally infringes all limitations of claim 1 except for the “pharmaceutically acceptable fluid” limitation. Slayback uses ethanol instead of propylene glycol (PG) in its generic product. Eagle argued that the ethanol in Slayback’s product is insubstantially different from the propylene PG in the claimed composition and, accordingly, Slayback’s product infringes under the doctrine of equivalents.
Slayback moved for a judgment of non-infringement on the pleadings under Federal Rule of Civil Procedure 12(c). Slayback argued that the disclosure-dedication doctrine barred Eagle’s claim of infringement under the doctrine of equivalents because the asserted patents disclose, but do not claim, ethanol as an alternative solvent to PG. Eagle counter-argued that the disclosure-dedication doctrine does not apply here because the asserted patents do not disclose ethanol as an alternative to PG for the claimed embodiment that contains an antioxidant. In fact, the specification only discloses ethanol when discussing unclaimed embodiments that contain chloride salt. Specifically, the asserted patents disclose three distinct “categories” of bendamustine formulations: (i) chloride salt formulations; (ii) antioxidant formulations; and (iii) dimethyl sulfoxide (“DMSO”) formulations. The specification only discloses ethanol as an alternative to PG when discussing the unclaimed chloride salt formulations; and it never discloses ethanol as an alternative to PG when discussing the claimed antioxidant formulations. Eagle also submitted an expert declaration from Dr. Mansoor Amiji in support of its opposition. Nevertheless, the district court held for Slayback because the asserted specification expressly and repeatedly identifies “ethanol” as an alternative “pharmaceutically acceptable fluid” to PG.
The federal circuit affirms the district court’s holding. The disclosure-dedication doctrine does not require the specification to disclose the allegedly dedicated subject matter in an embodiment that exactly matches the claimed embodiment. Instead, the disclosure-dedication doctrine requires only that the specification disclose the unclaimed matter as an alternative to the relevant claim limitation. That is, the disclosure-dedication doctrine requires disclosure of alternatives only at limitation level, not at embodiment level. In this case, the asserted patents disclose ethanol as an alternative to PG in the “pharmaceutically acceptable fluid” claim limitation. The specification repeatedly identifies—without qualification—ethanol as an alternative pharmaceutically acceptable fluid. Aside from the description of certain exemplary embodiments, nothing in the specification suggests that these repeated disclosures of ethanol are limited to certain formulations, or that they cannot extend to the claimed formulation.
Eagle also challenges the district court’s decision from the procedural grounds. Eagle argues that the district court erred by resolving that factual dispute at the pleading’s stage without drawing all reasonable inferences in Eagle’s favor. The federal circuit held that the district court has discretion to consider evidence outside the complaint for purposes of deciding whether to accept that evidence and convert the motion into one for summary judgment. In this case, the district court decided that Eagle was trying to fabricate a factual dispute and Slayback is entitled to judgment in its favor as a matter of law.
Take away
- The disclosure-dedication doctrine applies at limitation level, not at embodiment level.
- A patent drafter should be careful that the scopes of claims are consistent with the disclosure of specification to avoid inadvertent disclosure-dedication.
Tags: disclosure-dedication doctrine > doctrine of equivalent > patent infringement
One correct way of reciting a Markush group in the claim
| January 24, 2020
Amgen Inc. v. Amneal Pharmaceuticals LLC
January 7, 2020
Lourie (Opinion author), Newman, and Taranto
Summary
The Federal Circuit reversed the district court’s claim construction of the Markush groups as being closed to unrecited elements because the claim recites “comprising” as the transitional phrase and “at least one” in front of each of the Markush groups. Furthermore, the Federal Circuit affirmed the district court’s holding of noninfringement under the doctrine of equivalents by one of the Defendants’ products due to prosecution history estoppel.
Details
Amneal, Piramal, and Zydus each filed an Abbreviated New Drug Application (ANDA), seeking approval of a generic version of Sensipar®, a formulation of cinacalcet hydrochloride used to treat secondary hyperparathyroidism in adult patients with chronic kidney disease who are on dialysis and to treat hypercalcemia in patients with parathyroid cancer and primary and secondary hyperparathyroidism. Amgen holds the approved New Drug Application for Sensipar®, and sued them for infringing U.S. Patent No. 9,375,405 (the ‘405 patent).
The ‘405 patent is directed to a rapid dissolution formulation of cinacalcet. Claim 1 of the ‘405 patent is as follows:
A pharmaceutical composition comprising:
(a) from about 10% to about 40% by weight of cinacalcet HCl in an amount of from about 20 mg to about 100 mg;
(b) from about 45% to about 85% by weight of a diluent selected from the group consisting of microcrystalline cellulose, starch, dicalcium phosphate, lactose, sorbitol, mannitol, sucrose, methyl dextrins, and mixtures thereof,
(c) from about 1% to about 5% by weight of at least one binder selected from the group consisting of povidone, hydroxypropyl methylcellulose, hydroxypropyl cellulose, sodium carboxymethylcellulose, and mixtures thereof; and
(d) from about 1% to 10% by weight of at least one disintegrant selected from the group consisting of crospovid[o]ne, sodium starch glycolate, croscarmellose sodium, and mixtures thereof,
wherein the percentage by weight is relative to the total weight of the composition, and wherein the composition is for the treatment of at least one of hyperparathyroidism, hyperphosphonia, hypercalcemia, and elevated calcium phosphorus product.
The ‘405 patent issued from U.S. Patent Application 12/942,646 (the ‘646 application). The original ‘646 application contained only one claim, which recited a “pharmaceutical composition comprising an effective dosage amount of a calcium receptor-active compound and at least one pharmaceutically acceptable excipient, wherein” the composition achieved a specific dissolution profile. Later, Amgen filed a Preliminary Amendment, which cancelled claim 1 and added new claims 2-24. The newly filed claim 2, which ultimately issued as claim 1, is as follows:
A pharmaceutical composition comprising:
(a) from about 10% to about 40% by weight of cinacalcet HCl;
(b) from about 45% to about 85% by weight of a diluent selected from the group consisting of microcrystalline cellulose, starch, dicalcium phosphate, lactose, sorbitol, mannitol, sucrose, methyl dextrins, and mixtures thereof,
(c) from about 1% to about 5% by weight of at least one binder; and
(d) from about 1% to 10% by weight of at least one disintegrant,
wherein the percentage by weight is relative to the total weight of the composition.
The Examiner rejected the new claims. Then, Amgen filed an amendment of the independent claim 2, reciting “from about 10% to about 40% by weight of cinacalcet HCl in an amount of from about 20 mg to about 100mg.” Then, Amgen conducted a phone interview with the Examiner. During the interview, the Examiner proposed an Examiner’s Amendment of the independent claim 2, and Amgen accepted the Examiner’s Amendment. Specifically, the Examiner’s Amendment revised the binder and the disintegrant limitations into their current, Markush group format. Then, the claims are allowed. After the Notice of Allowance was issued, Amgen filed a number of RCE Requests with new IDS submission. In one of the RCE Requests, with regard to the previous Examiner’s Amendments, Amgen remarked that “[t]hese amendments have not been made in response to a prior art rejection but rather to place the claims in proper format and to better define the claimed subject matter, including equivalents.” The Examiner issued a Notice of Allowance after each RCE Request.
In the district court proceeding, one key issue is the construction of the binder and disintegrant Markush groups. The district court held that the Markush groups for the binder and disintegrant elements are closed to unrecited binders and disintegrants, relying on Multilayer Stretch Cling Film Holdings, Inc. v. Berry Plastics Corp., 831 F.3d 1350 (Fed. Cir. 2016). Furthermore, the district court held that Amneal does not infringe the ‘405 patent because Amneal uses Opadry Clear YS-1-7006 as the binder, a product that contains PEG 400 and PEG 8000 in addition to hydroxypropyl methylcellulose, which is a listed binder in claim 2. Furthermore, the district court held that Piramal does not infringe the ‘405 patent under the doctrine of equivalents because prosecution history estoppel barred Amgen’s argument that the cold-water soluble fraction of the preglatinized starch used by Piramal is equivalent to providone, a listed binder in claim 2.
On appeal, first, the Federal Circuit reversed the district court’s claim construction regarding the binder and disintegrant Markush groups. Specifically, contrary to the district court’s interpretation, because claim 1 uses “comprising” as the transitional phrase and “at least one” in front of each Markush group, the binder and disintegrant Markush groups do not preclude other binders and disintegrants in the claimed composition. Without more, such language is satisfied when an accused product contains a component that is from the Markush group and that meets the limitation’s other requirements such as concentration requirement for the component. Furthermore, the Federal Circuit held that Opadry used as a binder by Amneal contains HPMC, which is a listed binder in claim 1. Thus, irrespective of whether other components such as PEG are present, provided that Amneal’s formulation contains from about 1 to about 5% HPMC, the formulation literally infringes. Accordingly, the Federal Circuit vacates and remands those findings by the district court.
Next, the Federal Circuit held that Amgen’s doctrine of equivalents argument is barred by prosecution history estoppel. During the prosecution, Amgen accepted the Examiner’s Amendment that revised the claim’s disintegrant and binder limitations to be in Markush group format. Such an amendment narrows the scope of the claim, and Amgen failed to carry its burden to demonstrate that the Examiner’s Amendment was made for a reason unrelated to patentability. The Amgen’s statement in the later RCE Request was made over eight months after the Examiner’s Amendment was accepted, and accordingly does not provide any insight on the reasons for the Examiner’s Amendment. Furthermore, the tangential exception to prosecution history estoppel does not apply here because the use of pregelatinized starch as a binder is taught in the cited prior art. An amendment made to avoid prior art that contains the equivalent in question is not tangential.
Accordingly, the Federal Circuit vacated and remanded the district court’s judgment that Amneal does not infringe the ‘405 patent, and affirmed the district court’s judgment that Piramal does not infringe the ‘405 patent.
Take away
- The claims should recite “comprising” as the transitional phrase and “at least one” in front of the Markush groups to avoid being constructed to be closed to unrecited elements in the Markush groups.
- The original specification should contain a complete set of claims instead of one claim because later amendments may inevitably invoke prosecution history estoppel, which would bar most of the doctrine of equivalents arguments.
- The patent drafter should be mindful of an independent claim with an overly broad scope. Any later amendments during the patent prosecution may invoke prosecution history estoppel. Accordingly, the applicants have the risk of losing any protection afforded by patent infringement under the doctrine of equivalents.
WHEN FIGURES IN A DESIGN PATENT DO NOT CLEARLY SHOW AN ARTICLE OF MANUFACTURE FOR THE ORNAMENTAL DESIGN, THE TITLE AND CLAIM LANGUAGE CAN LIMIT THE SCOPE OF THE DESIGN PATENT
| October 7, 2019
Curver Luxembourg, SARL, v. Home Expressions Inc.
September 12, 2019
Chen (author), Hughes, and Stoll
Summary:
The Federal Circuit affirmed the district court’s grant of a defendant’s motion to dismiss a complaint for failure to state a plausible claim of design patent infringement because when all of the drawings in a design patent at issue do not describe an article of manufacture for the ornamental design, the title, claim language, figure descriptions specifying an article of manufacture, which was amended during the prosecution of the patent based on the Examiner’s proposed amendment, can limit the scope of a design patent.
Details:
The ‘946 Patent
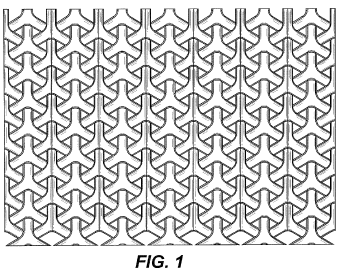
Curver Luxembourg, SARL (Curver) is the assignee of U.S. Design Patent No. D677,946 (‘946 patent) with a title “Pattern for a Chair” and claiming an “ornamental design for a pattern for a chair.” The ‘946 patent claims an overlapping “Y” design, as shown below. However, none of the figures illustrate a design being applied to a chair.
Prosecution
Curver originally applied for a patent directed to a pattern for “furniture,” and the original title was “FURNITURE (PART OF-).” The original claim recited a “design for a furniture part.”
However, during the prosecution, the Examiner allowed the claim but objected to the title because it was too vague to constitute an article of manufacture (in Ex Parte Quayle Action). The Examiner suggested amending the title to read “Pattern for a Chair,” and Curver accepted the Examiner’s suggestion by replacing the title with “Pattern for a Chair” and amending the claim term “furniture part” with “pattern for a chair.” Curver did not amend the figures to illustrate a chair. The Examiner accepted Curver’s amendments and allowed the application.


District Court
Home Expressions makes and sells baskets with a similar overlapping “Y” design disclosed in the ‘946 patent.
Curver sued Home Expressions accusing its basket products of infringing the ‘946 patent. Home Expressions filed a motion to discuss Curver’s complaint under Rule 12(b)(6) for failing to set forth a plausible claim of infringement.

Using a two-step analysis, the district court construed the scope of the ‘946 patent to be limited to the design pattern illustrated in the figures as applied to a chair and found that an ordinary observer would not purchase Home Expressions’s basket with “Y” design believing that the purchase was for “Y” design applied to a chair.
Therefore, the district court granted the Rule 12(b)(6) motion.
CAFC
The CAFC held that to define the scope of a design patent, the court traditionally focused on the figures illustrated in the patent. However, when all of the drawings fail to describe an article of manufacture for the ornamental design, the CAFC held that claim language specifying an article of manufacture can limit the scope of a design patent.
In addition, the CAFC uses §1.153(a) to held that “the design be tied to a particular article, but this regulation permits claim language, not just illustration along, to identify that article.”
The title of the design must designate the particular article. No description, other than a reference to the drawing, is ordinarily required. The claim shall be in formal terms to the ornamental design for the article (specifying name) as shown, or as shown and described.
The CAFC held that the prosecution history shows that Curver amended the title, claim, figure descriptions to recite “pattern for a chair” in order to satisfy the article of manufacture requirement necessary to secure its design patent. Therefore, the CAFC held that the scope of the ‘946 patent should be limited by those amendments.
Therefore, the CAFC affirmed the district court’s grant of Home Expressions’s motion to dismiss the complaint for failure to state a plausible claim of design patent infringement.
Takeaway:
- When figures in a design patent do not clearly show an article of manufacture for the ornamental design, the title and claim language can limit the scope of the design patent.
- Applicant should review the Examiner’s proposed amendments carefully before placing the application in condition for allowance.
- Applicant should be careful when crafting the title and claim language.
Tags: article of manufacture > claim > Design Patent > figure > patent infringement > title
The Federal Circuit indirectly imports limitations from the specification into the claims through claim construction
| April 24, 2017
The Medicines Company v. Mylan Inc. Etc.
April 6, 2017
Before Dyk Wallach, and Hughes. Opinion by Dyk.
Summary
Mylan submitted two Abbreviated New Drug Applications (“ANDA”) to seek approval of Food and Drug Administration (“FDA”) for selling generic bivalirudin drug products before the expiration of patents-in-suit: the ‘727 patent and the ‘343 patent. In its ANDA, Mylan stated that it would limit the Asp9 level of its generic product to less than 2.0 percent. Medicines sued Mylan for infringement of the ‘727 and ‘343 patents. Mylan filed counter-claims seeking declaratory judgments of invalidity. The district court held that Mylan infringed the ‘727 patent, but not the ‘343 patent. The Federal Circuit reverses in part, and held that Mylan does not infringe both patents.
A guide for the international patent attorney: how to read the recent CAFC en banc decision on laches–a viable defense against patent infringement, for the time being
| October 19, 2015
SCA Hygiene Products Aktiebolag v. First Quality Products, LLC
September 18, 2015
Introductory note from the author of this review: When I read this decision, it struck me that the legal analysis is based on concepts and distinctions which may not be clearly appreciated by someone not familiar with the “common law” tradition, the co-existence of remedies “at law” and remedies in “equity”, and the underlying principle of “separation of powers” in the federal government in the U.S. This en banc decision is very important, so there have been already numerous reports on the decision. I hope to contribute, in the form of the interspersed “notes” below, background information which may shed more light on the decision for our international colleagues. Le-Nhung McLeland
En banc decision: Opinion for court by Prost, joined by Newman, Lourie, Dyk, O’Malley, and Reyna. Opinion concurring-in-part, dissenting-in-part by Hughes, joined by Moore, Wallach, Taranto, and Chen. Judge Stoll did not participate in decision.
Note: Twenty three “amicus curiae” briefs were received in this case, which is a measure of its importance. The briefs were submitted on behalf of patent professional associations, as well as a broad range of corporations including Hewlett-Packard, Intel Corporation, Xerox Corporation, Johnson & Johnson, AT&T Mobility II LLC, T-Mobile USA, Roche Molecular Systems, Harley-Davidson Motor Company, and Rockwell Automation, Inc. The en banc majority refers to some of these briefs, in the body of the opinion or in footnotes.
Laches: a viable defense against patent infringement, but for how long?
| October 16, 2014
SCA Hygiene Products Aktiebolag v. First Quality Products, LLC
Summary:
SCA owns U.S. Patent No. 6,375,646 (the ‘646 patent) for adult incontinence products.
October 31, 2003: SCA sent a warning letter to First Quality, stating that SCA believes First Quality’s pants-type diapers infringe the ‘646 patent, and asking First Quality to either explain why the products do not infringe, or give assurance that First Quality will stop making and selling the products if First Quality believes the products to infringe the patent.
November 21, 2003: First Quality responded by letter, stating that prior art U.S. Patent No. 5,415,649 (the ‘649 prior art patent) invalidates the ‘646 patent.
July 7, 2004: SCA filed a request for ex parte reexamination of the ‘646 patent over the ‘649 prior art patent, but did not notify First Quality about the reexamination.
March 27, 2007: The PTO confirmed the patentability of all the original claims of the ‘646 patent and also granted new claims added during the reexamination.
August 2, 2010: SCA filed suit against First Quality for infringement of the ‘646 patent (six years and nine month after sending the warning letter, and more than three years after the conclusion of the reexamination.) First Quality counterclaimed for declaratory judgment of non-infringement and invalidity. After the district court’s claim construction order, First Quality moved for partial summary judgment of non-infringement, and summary judgment for laches and equitable estoppel. The district court granted summary judgment for laches and equitable estoppel, and dismissed the remaining motion as moot. SCA appealed.
The conclusion about laches or equitable estoppel is “committed to the sound discretion of the trial judge.” Therefore, the standard of review of those conclusions by the CAFC is “abuse of discretion” by the trial judge. Since the underlying elements of laches and equitable estoppel are questions of fact, the CAFC reviews those findings of fact for “clear error.” However, when summary judgment has been granted, the CAFC reviews the factual elements de novo to determine whether any “genuine issues of material fact remain.”
