No obviousness where hindsight argument relied upon “cherry-picked” data
| September 11, 2019
Sanofi-Aventis et al. v. Dr. Reddy’s Laboratories et al.
August 14, 2019
Lourie, Moore, Taranto. Opinion by Lourie
Summary
The CAFC upheld the nonobviousness of claims where the lead compound was known, but no cited art would have motivated the skilled artisan to arrive at the claimed compound. The claimed compound was a specific modification of the lead compound, and no analogous modification was found in the art. The CAFC rejected “cherry-picked” data which formed a “convoluted” obviousness argument, and stated that there is no per se rule that a compound is obvious where there is only a small change from a lead compound.
Details
Technical background
Sanofi owns U.S. Patent Nos. 5,847,170 and 8,927,592, which claim cabazitaxel (commercially known as Jevtana) and methods of use thereof. This drug is used to treat drug-resistant prostate cancers. Cabazitaxel is only the third taxane drug to obtain FDA approval, since the first being in 1992. Cabazitaxel is similar to docetaxel (approved in 1996) as shown below:
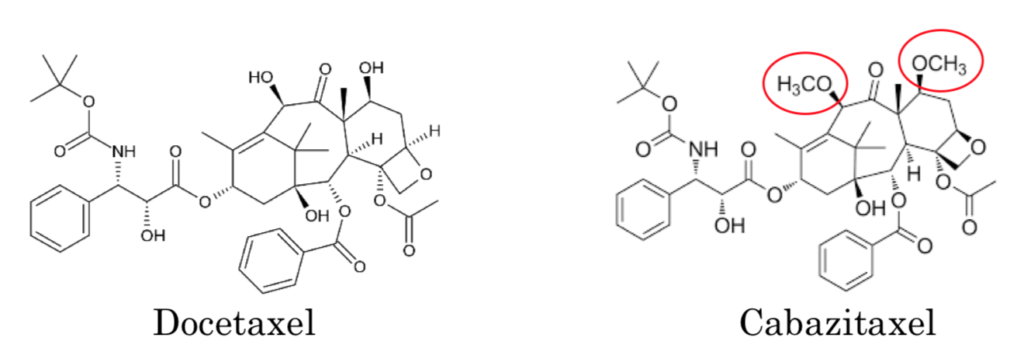
Rather than hydroxyl groups at the C7 and C10 positions, Cabazitaxel has methoxy groups.
Procedural background
The Defendants filed an ANDA alleging that the ‘592 and ‘170 patents are invalid as being obvious. While the district court case arising out of the ANDA litigation was pending, the PTAB instituted an IPR of the ‘592 patent. The PTAB found some claims 1-5 and 7-30 of the ‘592 patent unpatentable as being obvious and denied Sanofi’s motion to amend. Sanofi appealed the denial of the motion to amend (this has been remanded to PTAB in another CAFC case from earlier in 2019), but did not appeal the invalidity decision with respect to claims 7, 11, 14-16 and 26. Sanofi filed a statutory disclaimer with respect to these claims.
Back at the district court, even though the statutory disclaimer had been filed and the district court was notified, the court held that a case or controversy still existed with respect to the disclaimed claims, and held that they were obvious. The court also held that claims 1 and 2 of the ‘170 patent were not invalid as being obvious. Sanofi appealed the district court’s conclusion that a case or controversy exists with respect to the claims of the ‘592 patent, and some of the defendants appealed the district court’s decision regarding the lack of obviousness of claims 1 and 2 of the ‘170 patent.[1]
District Court
The defendants argued that cabazitaxel would have been obvious in view of docetaxel as a lead compound. The district court based its conclusion of non-obviousness on seven witnesses and 17 prior art references. The court found that a person skilled in the art would have been motivated to use docetaxel as a lead compound, but would not have been motivated to replace the C7 and C10 hydroxyl groups with methoxy groups to arrive at cabazitaxel.
The defendants alleged that it would have been obvious to modify docetaxel by increasing lipophilicity, in order to interfere with a protein called Pgp, which is involved in drug resistance. The defendants cited to two references, Hait and Lampidis.
In Hait, the authors studied phenothiazines, not taxanes. They showed that by increasing lipophilicity, the cancer cell would be more sensitive to the drug. However, this related to a different class of drugs, which are structurally very different from taxanes. Also, Hait only disclosed hypothetical binding models with respect to Pgp.
In Lampidis, the authors found that increasing the lipophilicity of a positively charged dye caused accumulation in drug resistant cells. But, Lampidis also never disclosed taxanes. Further, taxanes do not have a positive charge, unlike the dye of Lampidis.
Next, the district court considered a reference (Commercon) that identified the C2’, C3’, C7, C9 and C10 positions of paclitaxel as “flexible” and suitable for modification, in combination several other references regarding the activity of taxane analogs. Cumulatively, the district court found that the defendants had “cherry-picked” data from the references. The district court was persuaded by Sanofi’s expert who explained that taxane modifications had been considered at positions C2, C4, C5, C7, C8, C9, C10, C11, C12, C13, C14, C2’ and C3’, but that it would not have been obvious to modify just C7 and C10 to be methoxy groups.
The district court also found secondary considerations. Despite significant efforts by others, cabazitaxel was only the third taxane to get FDA approval. Furthermore, Jevtana achieved significant commercial success.
CAFC
At the CAFC, the defendants argued that that the district court erred, and that one skilled in the art (1) would have been motivated to modify docetaxel to decrease Pgp-related drug resistance, (2) knew that this could be done by increasing lipophilicity at the C7 and C10 positions, and (3) knew that the methoxy substitutions were the most conservative modifications.
However, the CAFC agreed with Sanofi that this obviousness theory is based on hindsight and said that it is “convoluted.” First, the CAFC indicated that Hait and Lampidis would not have provided a reason to make docetaxel more lipophilic, for the same reasons as the district court. But, even if there was a reason to make docetaxel more lipophilic, the cited art was highly varied in its suggested modifications of the lead compound. No single reference taught any type of simultaneous change at the C7 and C10 positions, and certainly not methoxy substitutions.
As to the Commercon reference, the defendants argued that C7, C9 and C10 are taught to be flexible, but that C3’ and C2’ were “crucial.” However, the text of the reference itself stated that C3’ is flexible and C2’ can be modified under certain conditions.
Finally, the defendants argued that even though no reference shows only the C7 and C10 methoxy substitutions, this would have been obvious because they are known to be small, conservative changes that increase lipophilicity. They pointed to a methylthiomethoxy substitution as also suggesting a methoxy substitution. The CAFC refuted this as hindsight, since there was no evidence that these two types of substitutions would behave similarly.
The defendants also argued that where there are a small number of changes to try, it is obvious to make a single change to a lead compound, particularly where the change is known to have desirable properties. Again, there was no showing of beneficial properties from methoxy substitutions of C7 and C10 in a taxane. The CAFC also stated that small changes are not prima facie obvious.
Finally, the CAFC found no clear error with the secondary considerations and affirmed the district court analysis of non-obviousness of the claims.
Takeaway
-If an obviousness argument relies on data that can be fairly characterized as “cherry picked,” odds are it is an unsuccessful hindsight argument.
-There
is no per se rule that a “small change” from a lead compound results in a
conclusion of obviousness.
[1] This decision also includes an extensive discussion of why the district court did not have authority to hold that there is a case or controversy regarding disclaimed claims. Discussion of that issue is omitted here.
